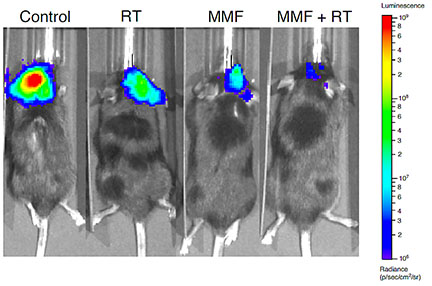
Purine metabolism regulates DNA repair and therapy resistance in glioblastoma
Intratumoral genomic heterogeneity in glioblastoma (GBM) is a barrier to overcoming therapy resistance. Treatments that are effective independent of genotype are urgently needed. Dan Wahl, MD, PhD and colleague’s recently published their work correlating intracellular metabolite levels with radiation resistance across dozens of genomically-distinct models of GBM, in which they found that purine metabolites, especially guanylates, strongly correlate with radiation resistance. Inhibiting GTP synthesis radiosensitizes GBM cells and patient-derived neurospheres by impairing DNA repair. Likewise, administration of exogenous purine nucleosides protects sensitive GBM models from radiation by promoting DNA repair. Neither modulating pyrimidine metabolism nor purine salvage has similar effects. An FDA-approved inhibitor of GTP synthesis potentiates the effects of radiation in flank and orthotopic patient-derived xenograft models of GBM. High expression of the rate-limiting enzyme of de novo GTP synthesis is associated with shorter survival in GBM patients. These findings indicate that inhibiting purine synthesis may be a promising strategy to overcome therapy resistance in this genomically heterogeneous disease.
Study Explains Why Patients with Cancer Spread to the Liver Have Worse Outcomes
The liver siphons critical immune cells to render immunotherapy ineffective; radiation to the liver may block this process. Metastasis is the primary cause of cancer mortality, and cancer frequently metastasizes to the liver. It is not clear whether liver immune tolerance mechanisms contribute to cancer outcomes. Mike Green, MD, PhD and colleagues reported in a recently published article that liver metastases diminish immunotherapy efficacy systemically in patients and preclinical models. Patients with liver metastases derive limited benefit from immunotherapy independent of other established biomarkers of response. In multiple mouse models, they show that liver metastases siphon activated CD8+ T cells from systemic circulation. Within the liver, activated CD8+ T cells die, consequently, creating a systemic immune desert. Similarly, patients with liver metastases have reduced peripheral T cell numbers and diminished tumoral T cell diversity and function. In preclinical models, liver-directed radiotherapy eliminates immunosuppressive hepatic macrophages, increases hepatic T cell survival and reduces hepatic siphoning of T cells. Thus, liver metastases co-opt host peripheral tolerance mechanisms to cause acquired immunotherapy resistance through CD8+ T cell deletion, and the combination of liver-directed radiotherapy and immunotherapy could promote systemic antitumor immunity.
Yu, J., Green, M.D., Li, S. et al. Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. Nat Med (2021). https://doi.org/10.1038/s41591-020-1131-x

Improving Quality and Consistency in Clinical Trials via Knowledge-Based Planning: 2017 Best of ASTRO
Consistency and standardization of radiotherapy plan quality in multi-institutional clinical trials are always challenging, and non-protocol compliant radiotherapy plans have been shown to impact patient outcomes. We performed a study aimed at employing knowledge-based planning (KBP) as a method of producing high quality, consistent, protocol-compliant treatment plans, in a complex setting of spinal stereotactic body radiation therapy (SBRT) on NRG Oncology RTOG 0631. Our KBP model generated plans meeting all protocol objectives in a single optimization when tested on both internal and external RTOG 0631 cases. Two external plans submitted under the protocol that were considered to have a protocol-unacceptable deviation were made protocol compliant through the use of the model. We believe that incorporation of KBP models into the clinical trial setting may have a profound impact on the quality of trial results due to the increase in consistency and standardization of planning, especially for treatment sites or techniques that are non-standard. This work was supported by NRG Oncology, the National Cancer Institute, and Varian Medical Systems, Inc.
KC Younge, R Marsh, D Owen, H Geng, Y Xiao, DE Spratt, J Foy, QRJ Wu, FF Yin, S Ryu, MM Matuszak, 2017 Best of ASTRO
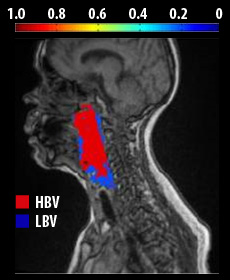
Using advanced radiologic imaging to increase radiation doses to resistant parts of the tumor
In order to overcome resistance to therapy in poor- prognosis cancer of the head and neck we use advanced imaging called functional MRI (FMRI) to detect parts of the tumor that are expected to be more resistant, including the parts which have less blood supply and parts that have a higher concentration of tumor cells. Once we detect these parts of the tumor, we then plan a higher dose of radiation to be delivered to these parts. This results in a potential for a better eradication of the tumor without increasing complication rates (which would be expected had we increased the radiation doses to the whole tumor). This study is funded by the National Cancer Institute.
Tomography, 2(4):341-352, 2016, You Dk, Aryal M, Samuels SE, Eisbruch A, Cao,Y