Expanded Prostate Cancer Index Composite
What is EPIC?
The Expanded Prostate Cancer Index Composite (EPIC) is a comprehensive instrument designed to evaluate patient function and bother after prostate cancer treatment. Content from the original UCLA-Prostate Cancer Index (PCI) was expanded with guidance from a development cohort of localized prostate cancer patients and an expert panel comprised of urological oncologists, radiation oncologists (including brachytherapy expertise), survey researchers, and prostate cancer nurses. These experts, patients, and review of the literature suggested a need to augment the UCLA-PCI with items to capture additional concerns relevant to brachytherapy, external beam radiation, radical prostatectomy, and androgen deprivation. Accordingly, the UCLA-PCI was supplemented with specific items addressing irritative and obstructive voiding symptoms (the original UCLA-PCI had queried principally incontinence only in urinary function assessment), hematuria, additional bowel symptoms (to improve the suboptimal bowel function scale from the original UCLA-PCI) and hormonal symptoms. Symptom-specific bother items corresponding to each symptom item were added to elicit multi-item bother scales for each HRQOL domain. Responses and comments from the development cohort were incorporated to derive the final instrument: the Expanded Prostate cancer Index Composite (EPIC).
EPIC Instruments
- Most Recent Version of EPIC
- 26-item short-form version of EPIC
- EPIC + SF12 and AUASI
- EPIC - Urinary Domain only
- EPIC - Bowel Domain only
- EPIC - Sexual Domain only
- EPIC - Hormonal Domain only
- EPIC - Demographics add-on*
*This instrument can be added on to the end of any of the other EPIC instruments to collect demographic information.
Peer-reviewed Articles
Scoring Utilities
- Scoring Documentation
- Scoring Instructions for the EPIC-26
- SAS Macro for scoring EPIC*
- SAS Macro for scoring EPIC-26*
* Choose the 'Save to Disk' option after clicking on the SAS Macro name. If your browser displays the macro on the screen as a text file, choose File -> Save As, and then choose a location to save the macro.
Minimally Important Differences (MID) for EPIC-26
* Based on pooled averages from distribution- and anchor-based approaches.
Sample Size Table & Suggested Endpoint
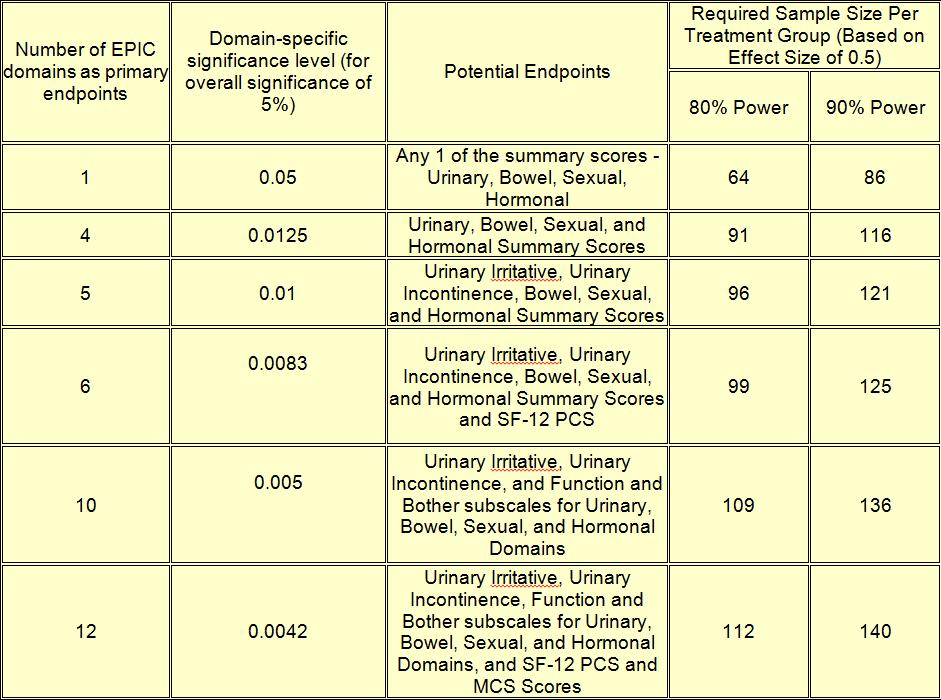
If less than 100% response rate is anticipated, then sample sizes will need to be adjusted. For example, an expected 75% response rate at 2 years for a study with a single endpoint and 80% power would require 86 subjects (64/0.75 = 85.33)
Translations to non-English languages and dialects
Control-based Norms and Other Statistics
Validation of EPIC-26 (EPIC Short Form)
Statistics for the validation process of the EPIC-26
Frequently Asked Questions
What does EPIC stand for? EPIC stands for the Expanded Prostate Cancer Index Composite.
What is EPIC? EPIC is a comprehensive instrument designed to evaluate patient function and bother after prostate cancer treatment.
Am I allowed to make copies of EPIC? Yes, you are free to make copies and distribute EPIC.
Do I need to pay a fee to use EPIC? No, there is no fee required to use EPIC.
Is EPIC available in any languages or dialects other than US English? Yes, please go to EPIC Translation Repository for non-English versions of EPIC.
When reporting my findings, what article should I reference? For any manuscript published that uses EPIC, you should reference the EPIC validation paper published in the December 2000 issue of Urology. The full reference is:
Wei J, Dunn R, Litwin M, Sandler H, and Sanda M. "Development and Validation of the Expanded Prostate Cancer Index Composite (EPIC) for Comprehensive Assessment of Health-Related Quality of Life in Men with Prostate Cancer", Urology. 56: 899-905, 2000.
Contact Information
Martin G. Sanda, M.D. Department of Urology
Emory University School of Medicine
201 Dowman Drive
Atlanta, GA 30322
Phone: (404) 727-6123
[email protected]
John T. Wei, M.D. University of Michigan
Department of Urology
3875 TC
1500 E. Medical Center Drive
Ann Arbor, MI 48109-5759
Phone: (734) 615-3040 Fax: (734) 936-9536
[email protected]
Mark S. Litwin, M.D., M.P.H. UCLA Department of Urology
Box 951738
Los Angeles, CA 90095-1738
Phone: (734) 615-3040 Fax: (734) 936-9127
[email protected]
For information on the use of the SAS scoring macro, please contact:
Rodney L. Dunn, M.S. University of Michigan
Department of Urology
2800 Plymouth Rd NCRC B16-150S
Ann Arbor MI 48109-2800
Phone: (734) 615-1396 Fax: (734) 936-9536
[email protected]

